Powering smarter, patient-centric clinical trials
United in medicine.
Expanding the potential of evidence generation
uMed Solutions
The two foundations of uMed
At uMed, we believe the best breakthroughs come when patient experience meets rigorous data. Our mission is to enable faster, smarter research by connecting consented patients, verified clinical datasets and agile study design — so therapies reach the people who need them, sooner.

Cohorts
Our ACCESS Cohorts are built to be a living research resource — reusable, scalable and designed for real-world impact. By linking consented patients with their routine care data and enabling remote engagement, we unlock faster, more representative evidence generation. Whether you’re exploring unmet patient needs, designing a trial or generating post-launch outcomes, our cohorts are ready when you are.
Learn more
Study Services
Support your upcoming study with uMed's modular suite of decentralised services. From recruitment, data collection, sampling and study management, uMed can design studies that centralise the patient and maximise the quality and quantity of end points collected.
Learn moreHow we help
End evidence for every stage of drug development
At uMed, we believe the best breakthroughs come when patient experience meets rigorous data. Our mission is to enable faster, smarter research by connecting consented patients, verified clinical datasets and agile study design — so therapies reach the people who need them, sooner.

Pharma teams validating launch strategy
You need real-world truth, not isolated signals. Traditional qualitative insight misses clinical grounding and diverse representation.
uMed Delivers:

Patient-reported experience linked directly to EMR/EHR.

Access to diverse, real-world cohorts beyond sites.

Integrated evidence for positioning, segmentation, and launch planning.

Clinical teams running pragmatic studies
You need confident delivery with strong retention and clear visibility.
uMed Delivers:

End-to-end decentralised trials without site burden.

Active oversight of the full patient journey with a single-patient-view from recruitment to completion.
Clinical trial teams recruiting patients
Timelines slip when eligible patients are slow to find, sites overload, and enrolment narrows.
uMed Delivers:

Rapid access to pre-consented, EMR-verified cohorts ready to activate.

Remote, low-burden participation for faster, broader recruitment.

Automated screening, consent, and follow-up to prevent site bottlenecks.
Some of our key partners
We are working with top institutions & vendors across the US & UK




















































































Case Studies
Real-time, technology enabled data collection ensures that you have the most up-to-date insights, minimizing gaps and errors in data.

RSV
RSVoice
A study evaluating the safety and immune response of an investigational vaccine aimed at preventing RSV infection in high risk adults.
Read more
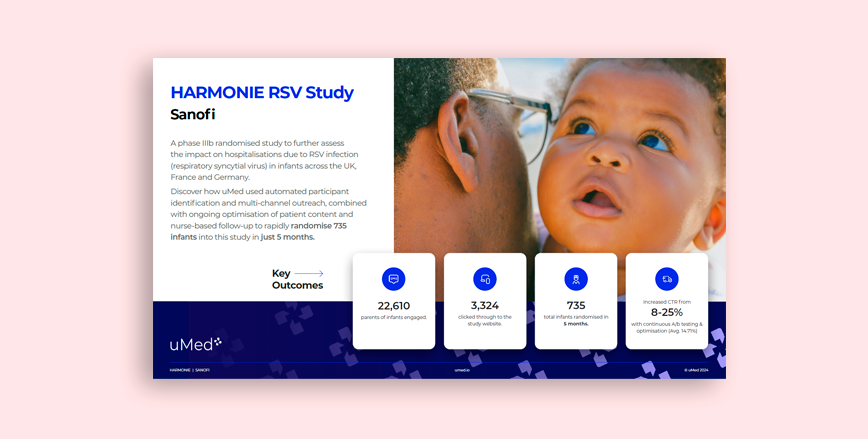
Paediatrics
HARMONIE
A study to assess the impact on hospitalisations due to RSV infection (respiratory syncytial virus) in infants across the UK, France and Germany.
Read more
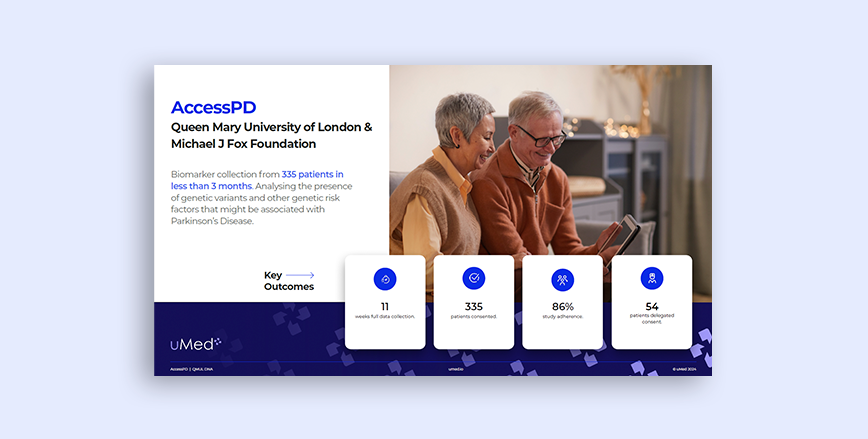
Parkinson's Disease
QMUL DNA
Discover how uMed facilitated biomarker collection from 335 patients in less than 3 months.
Read more
Partnerships
uMed Announces Strategic Partnership with WellSky® to Expand Patient Access to National Clinical Research Registries
Read the press releaseCompliance
Security-first recruitment that meets the highest regulatory standards.
At uMed we prioritise patient trust — ensuring privacy, transparency, and compliance every step of the way.

Fully ISO27001 and ISO9001 certified, Cyber Essentials Plus and NHS Digital Data & Security Toolkit certified.

HIPAA-compliant platform for cross-border trials.

Personal identifiable data (PII) is stored separately from health data — only encrypted link identifiers connect them.

Data use is strictly limited — only for approved studies with explicit patient consent. Any health data shared with researchers is always de-identified.

Patients are always in control: they can opt out at any time. No data is used for marketing or unrelated purposes.

















Get in touch
Book a capabilities demonstration