uMed Study Services
End-to-end Study Services, built for real-world research
uMed’s Study Services unite real-world patient engagement, multi-modal data capture, and high-quality recruitment to accelerate trials and generate robust evidence at scale.
Reach & scale
10M+ patients across UK & US
Speed
Faster recruitment and quicker delivery of clean datasets
Quality evidence
Validated PROs, devices, assays, and linked EHR data
Low burden
Pre-screened referrals, automated workflows, and nurse support
Compliance
IRB-approved, HIPAA & GDPR compliant
What are Study Services?
A smarter, scalable model for real-world & site-based research
uMed Study Services combine two powerful capabilities — Recruit and Collect — to support every stage of a study. From identifying eligible participants to capturing multi-source real-world data, our integrated workflows deliver speed, quality, and representativeness with less operational burden on sites, sponsors and patients.

A completely modular system of services
Address single trial challenges, or configure a complete trial
Recruitment
Highly qualified, site-ready patients identified through a rigorous three-stage screening process.
Decentralised, longitudinal data collection
Sustain data collection over time for sharper outcomes and richer insights - without keeping sites open.
Remote Trial Services
Conduct collection, treatment and interaction remotely or at the patients home or other convenient location rather than central sites for more resource-efficient, patient-centric trials.
Patient Support Hub
End-to-end centralised patient engagement lifecycle management, from enrollment and collection, through to communication and support, creates a unified patient experience, making participation easy and seamless.
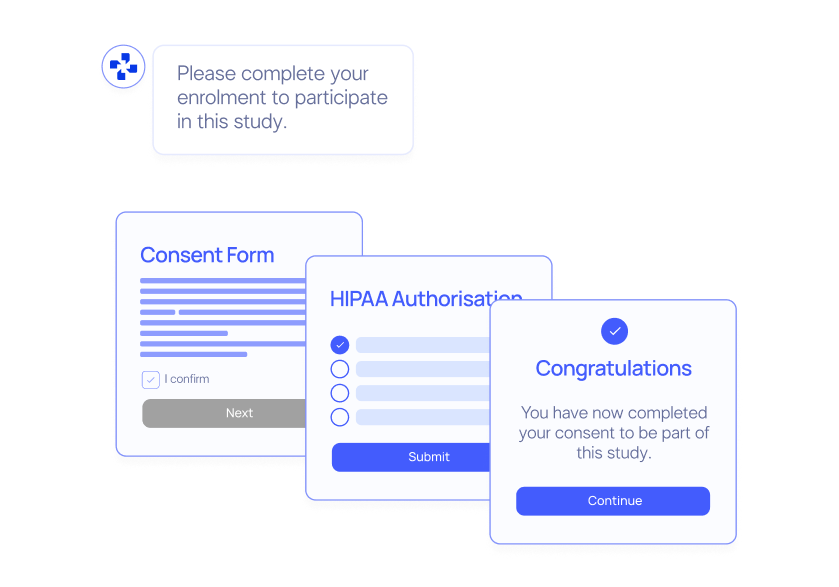
Remote Bio Sampling
Collect patient biological samples remotely, allowing for greater flexibility, convenience and data richness.
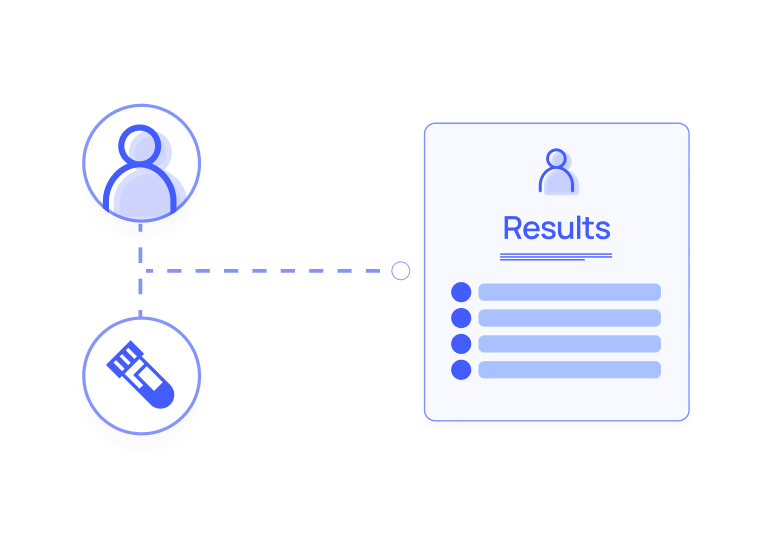
Smart Data Collection
Tech-enabled smart data collection (ePRO, EHR, primary & secondary care data) empowers faster, more convenient collection and consolidation of patient data required to support studies (including clinical, behavioral and demographic).
Study Service solutions

Fully decentralised
From identifying eligible participants to capturing multi-source, real-world data, our integrated workflows deliver speed and quality while keeping patients at the centre of the study.
Decentralised studies are designed and delivered around the patient experience, reducing burden on both sites and sponsors without compromising scientific rigour.
Decentralised studies are designed and delivered around the patient experience, reducing burden on both sites and sponsors without compromising scientific rigour.
Paediatric Vaccine trial
142
GPs participating as trial sites
1,100
Patients recruited and vaccinated in 8 weeks
3 month
Remote patient and EHR data collection follow up period

Recruit
Study Services Recruit accelerates trial enrolment by delivering highly qualified, site-ready patient referrals through clinically validated EHR identification, multi-channel outreach, and a rigorous three-stage screening process.
RSVoice - evaluating the safety and immuneresponse of an investigational vaccine
67
Referrals
17
Randomisations
25%
Conversion

Hybrid
Study Service's Hybrid studies combine uMed-led recruitment and remote data collection with in-person site visits, reducing operational burden for sites and sponsors while preserving the value of face-to-face clinical care.
MJFF PPMI - Supported identification, recruitment, and facilitated distribution of Smell Test to participants.
1200+
Physical ‘Smell Test’ kits deployed to patients.
400
Patients referred to sites.
How we work with you
Built for everyone involved in research


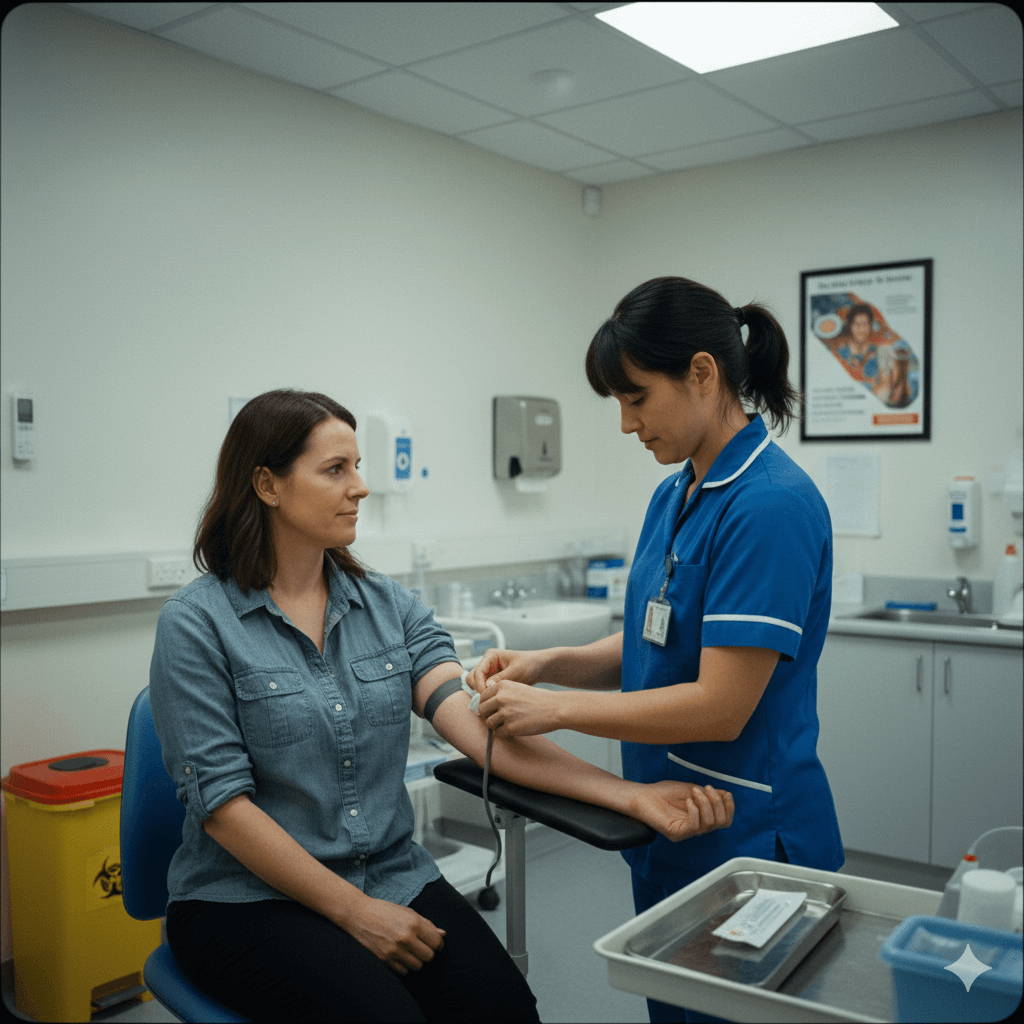
For Sponsors & CROs
Accelerate timelines, unlock real-world evidence, and reduce operational complexity.
For Sites & HCPs
Receive pre-screened referrals or support remote data collection — with almost no additional workload.
For Patients
Join studies from home, supported by nurse-led calls and user-friendly digital tools.
Study service solutions
Two services. Endless study possibilities.
Purpose
Identification
Data Capture
Output
Study Type
Site Burden
Data Depth
Timelines
uMed Recruit
Enroll patients into site-based trials
EMR + digital + nurse screening
Pre-screened referrals
Eligible, ready-to-enroll patients
Interventional
Low
Moderate
Faster enrollment
uMed Collect
Capture multi-source real-world data
Cohort-based consent + EMR
PROs, devices, assays, EHR
Publication-ready datasets
Observational, hybrid, post-market
None
High
Continuous evidence
Proven Impact
Delivering results for leading sponsors, sites & patients

Flusniff
Embedded one of the UK’s largest toddler flu-vaccine safety studies in NHS primary care — 1,920 consents across 147 practices in 7 weeks, enabling rapid IMP administration and high GP engagement.
1,920
Consents
147
Practices engaged
7 week
Timeline
95%
GP engagement

Start building with ACCESS
Book a complimentary demonstration